The Fallacy of Laboratory Reference Ranges
- Apr 3
- 5 min read
Why “Normal” Laboratory Values Often Fail to Represent Optimal Health
By Dr. Mark L. Gordon APR 2026

For decades, modern medicine has relied on a reassuring word to decide who needs help and who does not—that word is normal. If a blood test falls within a laboratory’s reference range, patients are often told everything is fine. Go home. Don’t worry. Come back next year.
But what if “normal” no longer means healthy?
Across doctors’ offices in the United States, millions of patients live with fatigue, brain fog, weight gain, depression, low libido, poor sleep, and declining resilience—despite being repeatedly told their laboratory results are unremarkable. Their blood work, after all, is “within range.” The problem is not imaginary. The problem may be the range itself.
How Reference Ranges Came to Rule Medicine
Laboratory reference ranges were never designed to define optimal health. They emerged in the mid-20th century as statistical tools, meant to flag extreme abnormalities. Most are built by sampling a broad population and defining “normal” as the middle 95% of values. If you fall inside that window, you pass.
This method assumes that the population itself is healthy enough to serve as a benchmark. That assumption may once have been reasonable. It is no longer.
Today’s reference populations include people who are sedentary, chronically inflamed, sleep-deprived, insulin resistant, hormonally depleted, stressed, medicated, and metabolically compromised. When such a population is averaged, the statistical middle no longer represents vitality. It represents mediocrity.
In effect, medicine has quietly redefined health as whatever is common.
The Difference Between Being Sick and Being Unwell
Most medical care is designed to detect disease, not to preserve function. This distinction matters. Disease is what happens when systems fail. Dysfunction begins long before that failure becomes obvious.
A patient may not meet criteria for hypothyroidism, hypogonadism, anemia, or inflammatory disease, yet still experience real physiological decline. Their hormones are technically “normal,” but insufficient for the demands of daily life. Their inflammation markers are acceptable, but high enough to impair cognition or mood. Their metabolism is compensating, but strained.
Medicine has a name for values that fall below the reference range: deficiency. It has no formal language for what happens before that line is crossed. Call it insufficiency. Call it suboptimal physiology. Call it the long slope before the cliff.
Most patients live on that slope for years.
Testosterone, Thyroid, and the Illusion of Reassurance
Consider testosterone. A man in his early fifties may present with fatigue, loss of muscle, low motivation, and diminished libido. His total testosterone comes back at 340 nanograms per deciliter—comfortably above the laboratory cutoff of 280. He is told he is normal.
Statistically, that is correct. Physiologically, it may be meaningless.
Testosterone does not function as a simple on–off switch. Its effects depend on receptor activation, tissue sensitivity, conversion to other neuroactive hormones, and interaction with inflammation and stress. Many men with “low-normal” levels experience real functional impairment, while those optimized into higher physiological ranges often report improved cognition, mood, and metabolic health.
Thyroid hormones tell a similar story. Most laboratories define a normal thyroid-stimulating hormone (TSH) range that extends well above levels associated with fatigue, weight gain, depression, and cardiovascular risk. Patients may hover in this gray zone for years, symptomatic but untreated, because their numbers do not yet violate statistical boundaries.
Inflammation markers complete the picture. A C-reactive protein level just below the upper limit of normal may still reflect ongoing vascular or neuroinflammation. Homocysteine levels deemed acceptable may quietly increase the risk of cognitive decline. These values are not benign simply because they are common.
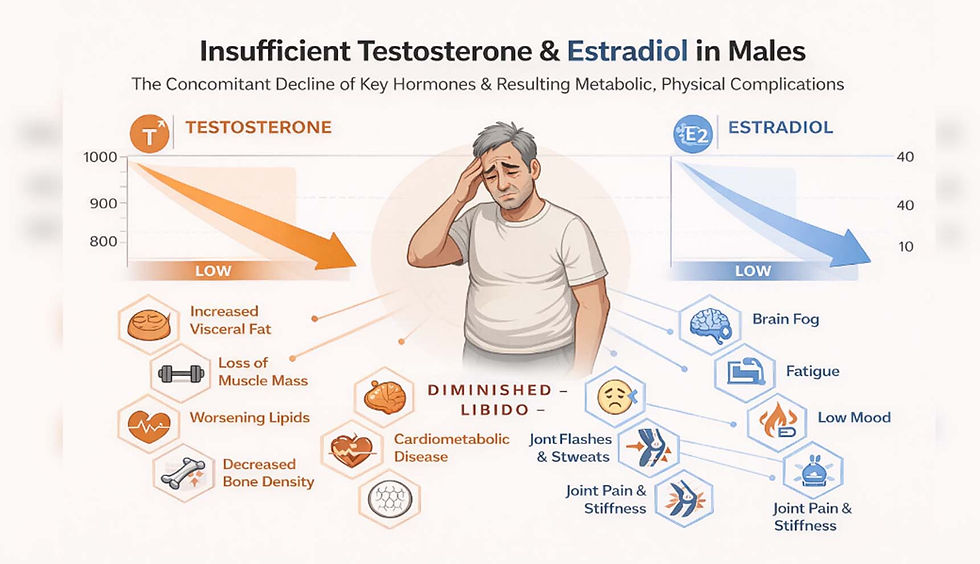
The Hidden Cost of “Within Range”
Biology is not static. When hormone levels drift lower, when nutrients thin out, when inflammation creeps upward, the body adapts. It compensates. Mitochondria work harder. Stress hormones rise. Repair mechanisms slow down.
Over time, compensation becomes exhaustion.
The brain is especially sensitive to this slow erosion. Suboptimal hormone and nutrient levels impair energy production, neurotransmission, and neuroplasticity. Mood disorders, cognitive fog, and reduced stress tolerance often appear long before imaging or traditional diagnostics reveal anything “wrong.”
By the time laboratory values cross into abnormal territory, years of silent wear have already occurred.
Why the System Keeps Missing It
Reference ranges are attractive because they are simple. They allow clinicians to sort results into binary categories: normal or abnormal. They protect against overdiagnosis. They are easy to defend.
But simplicity comes at a cost. Human physiology is networked, dynamic, and deeply individualized. A number that is adequate for one person may be insufficient for another, depending on genetics, age, stress exposure, inflammation, sleep, and environmental burden.
Treating lab values in isolation ignores these interactions. It treats numbers, not people.
A Shift Already Underway
A quiet shift is beginning in parts of medicine that deal with performance, longevity, and complex chronic illness. Instead of asking whether a value is normal, clinicians are asking whether it is working.
They track trends over time rather than single snapshots. They interpret hormones alongside inflammatory markers, nutrients, and symptoms. They establish personal baselines and ask what range supports energy, cognition, mood, and resilience for that individual.
In this model, health is not defined by population averages but by outcomes.
Technology is accelerating this transition. Advanced analytics and artificial intelligence now allow clinicians to detect subtle patterns across multiple biomarkers, often identifying risk trajectories long before disease manifests. Wearable devices and continuous monitoring are beginning to blur the line between laboratory testing and daily life.
The goal is no longer to wait for failure, but to preserve capacity.

Rethinking What “Normal” Means
The uncomfortable truth is that medicine has grown accustomed to managing decline. Reference ranges, as currently used, reflect that mindset. They tell us when someone is sick enough to intervene, not when they are drifting away from health.
But if the purpose of medicine is not merely to prolong life, but to preserve its quality, then statistical normality is an inadequate standard.
Health is not the absence of disease. It is the presence of adaptability—the ability to recover, recalibrate, and repair.
As populations age and chronic illness becomes the norm rather than the exception, the question is no longer whether reference ranges are scientifically valid. They are. The question is whether they are sufficient.
For millions of patients told they are “fine” while feeling anything but, the answer is already clear.
Disclaimer:
Contributor content reflects the personal views and experiences of the author and does not necessarily represent the views of Biohack Yourself Media LLC, Lolli Brands Entertainment LLC, or any of their affiliates. Content is provided for editorial, educational, and entertainment purposes only. It is not medical or dental advice. Always consult qualified professionals before making health decisions. By reading, you agree to hold us harmless for reliance on this material. See full disclaimers at www.biohackyourself.com/termsanddisclaimers


.jpg)

.jpg)











